Results for Grignard reaction:
•The theoretical yield for the Grignard reaction was 1.3092 g of 1-(4-methoxyphenyl)-3-buten-1-ol, while the actual yield turned out to be 0.7861 g. Therefore, the percent yield was calculated to be 60%.
After the reaction was complete and the product was purified, a proton NMR was taken of the 1-(4-methoxyphenyl)-3-buten-1-ol. The NMR shows that the product made was pure.
•The theoretical yield for the Grignard reaction was 1.3092 g of 1-(4-methoxyphenyl)-3-buten-1-ol, while the actual yield turned out to be 0.7861 g. Therefore, the percent yield was calculated to be 60%.
After the reaction was complete and the product was purified, a proton NMR was taken of the 1-(4-methoxyphenyl)-3-buten-1-ol. The NMR shows that the product made was pure.
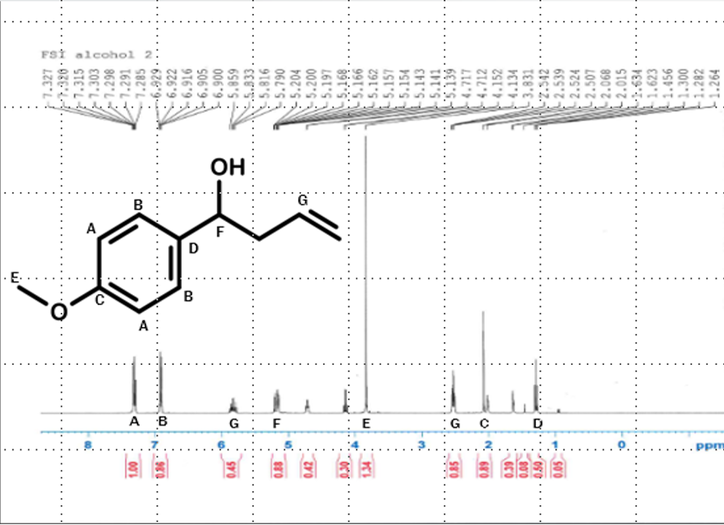
H-NMR scan indicating that the alcohol from the grignard reaction formed in the process.
Results for oxidation reaction:
•The theoretical yield for the oxidation reaction was 0.7771 g of 1-(4-Methoxyphenyl)-3-buten-1-one, while the actual yield was massed out to 0.6635 g. From this data, the percent yield was determined to be 75%.
Once the reaction had finished and the end product isolated, another proton NMR was taken, this time of the 1-(4-methoxyphenyl)-3-buten-1-one. This NMR also shows the compound to be fairly pure.
•The theoretical yield for the oxidation reaction was 0.7771 g of 1-(4-Methoxyphenyl)-3-buten-1-one, while the actual yield was massed out to 0.6635 g. From this data, the percent yield was determined to be 75%.
Once the reaction had finished and the end product isolated, another proton NMR was taken, this time of the 1-(4-methoxyphenyl)-3-buten-1-one. This NMR also shows the compound to be fairly pure.
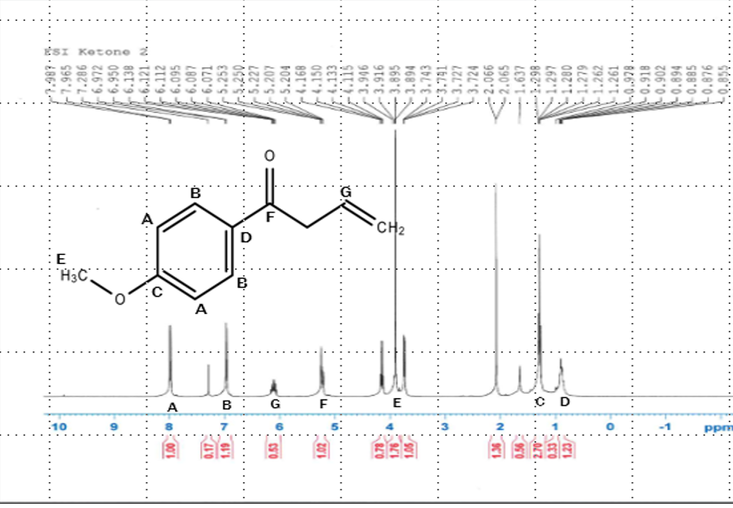
.H-NMR scan of ketone after oxidation reaction. Shows that a ketone was actually formed and the base for the isoxazole ring has started to form.
Results for oxime reaction:
•It was determined that the theoretical yield for the oxime reaction was 0.5818 g of (IE)-N-hydroxy-1-(4-methoxyphenyl)-3-buten-1-imine. The actual yield of the product was 0.4997 g, yielding 86% product.
•The oxime was filtered and purified, and a proton NMR was taken. The results showed the oxime to be pure as well.
•It was determined that the theoretical yield for the oxime reaction was 0.5818 g of (IE)-N-hydroxy-1-(4-methoxyphenyl)-3-buten-1-imine. The actual yield of the product was 0.4997 g, yielding 86% product.
•The oxime was filtered and purified, and a proton NMR was taken. The results showed the oxime to be pure as well.
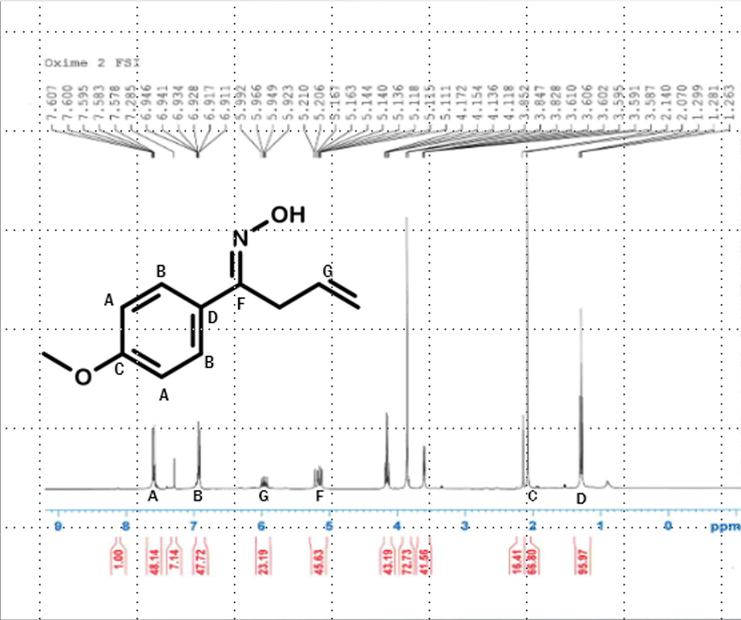
H-NMR scan after oxime reaction indicating that a pure form of the oxime was created indicating that it can be oxidized and that the isoxazole ring has continued to build meaning it can be completed.
Results for nickel (II) synthesis:
•The theoretical yield for the nickel synthesis was 0.153 g of the nickel-based 3,5-Disubstituted ∆2-isoxazoline, while the actual yield was 0.1098 g. The percent yield, therefore, was 72%. (However the reason the yield was so high was because of the solvent still left in the solution)
•After the isoxazoline was purified, a proton NMR was taken of it, and the results showed that little product had actually been made.
•The theoretical yield for the nickel synthesis was 0.153 g of the nickel-based 3,5-Disubstituted ∆2-isoxazoline, while the actual yield was 0.1098 g. The percent yield, therefore, was 72%. (However the reason the yield was so high was because of the solvent still left in the solution)
•After the isoxazoline was purified, a proton NMR was taken of it, and the results showed that little product had actually been made.
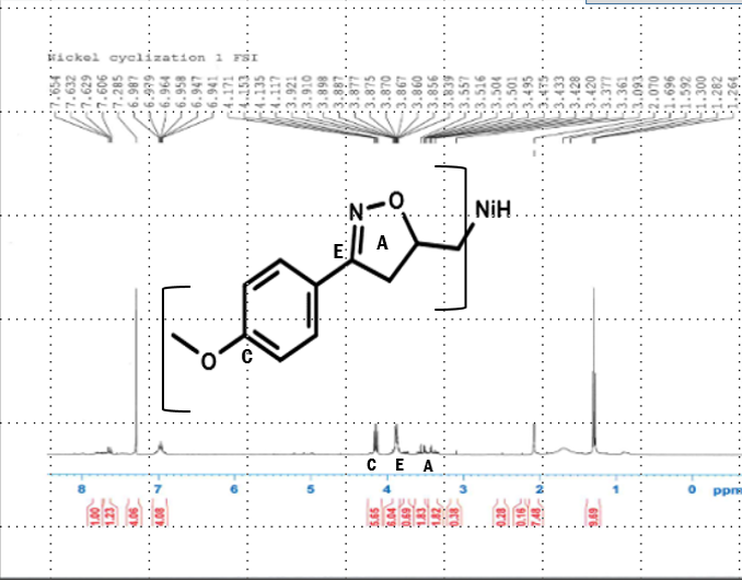
H-NMR of nickel-catalyzed isoxazoline showing that the nickel did not form the isoxazole ring as strong as expected indicating that it did form but not as well as other metalloids have.
Results for palladium (II) synthesis:
•The theoretical yield for the palladium synthesis was o.1796 g of the palladium-based 3,5-Disubstituted ∆2-isoxazoline, while the actual yield was 0.0796 g, yielding 44% product.
•The theoretical yield for the palladium synthesis was o.1796 g of the palladium-based 3,5-Disubstituted ∆2-isoxazoline, while the actual yield was 0.0796 g, yielding 44% product.
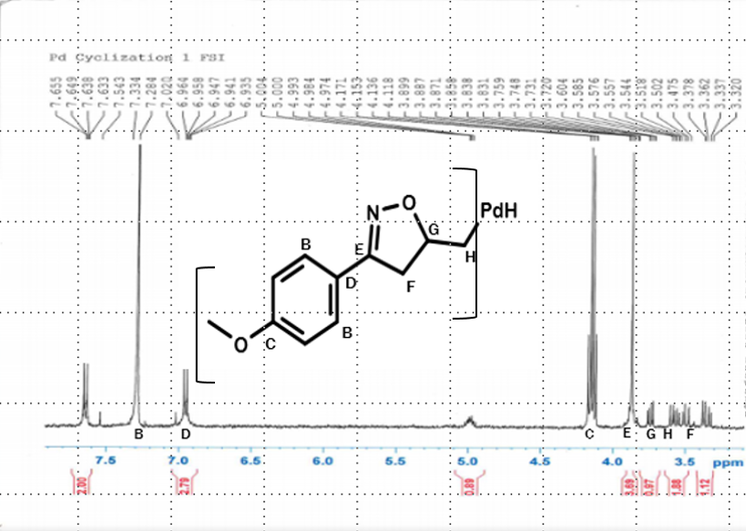
H-NMR of palladium-catalyzed isoxazoline indicating that the isoxazole ring actually formed and it formed stronger than than the nickel did.
